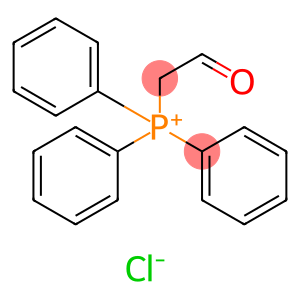
Formylmethyltriphenylphosphonium chloride
(formylmethyl)triphenylphosphonium chloride
CAS: 62942-43-2
Molecular Formula: C20H18ClOP
Formylmethyltriphenylphosphonium chloride - Names and Identifiers
| Name | (formylmethyl)triphenylphosphonium chloride |
| Synonyms | FMC LABOTEST-BB LT00233133 2-OXOETHYLTRIPHENYLPHOSPHONIUM CHLORIDE FORMYLMETHYLTRIPHENYLPHOSPHONIUM CHLORID Formylmethyltriphenylphosphonium chloride TRIPHENYLFORMYLMETHYLPHOSPHONIUM CHLORIDE (2-oxoethyl)(triphenyl)phosphonium bromide (formylmethyl)triphenylphosphonium chloride (FORMYLMETHYL)TRIPHENYLPHOSPHONIUM CHLORIDE (2-Oxoethyl)(triphenyl)phosphonium chloride Phosphonium, (2-oxoethyl)triphenyl-, chloride phosphonium, (2-oxoethyl)triphenyl-, hydrochloride |
| CAS | 62942-43-2 |
| EINECS | 263-767-2 |
| InChI | InChI=1/C20H18OP.ClH/c21-16-17-22(18-10-4-1-5-11-18,19-12-6-2-7-13-19)20-14-8-3-9-15-20;/h1-16H,17H2;1H/q+1 |
| InChIKey | RVEJRPJGKXTQIF-UHFFFAOYSA-M |
Formylmethyltriphenylphosphonium chloride - Physico-chemical Properties
| Molecular Formula | C20H18ClOP |
| Molar Mass | 340.78 |
| Melting Point | 209-212 °C (dec.) (lit.) |
| Water Solubility | Soluble in water. |
| Appearance | solid |
| Color | Light yellow to Yellow to Orange |
| BRN | 4060813 |
| Storage Condition | 2-8°C |
| Sensitive | Hygroscopic |
Formylmethyltriphenylphosphonium chloride - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R41 - Risk of serious damage to eyes R34 - Causes burns |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S39 - Wear eye / face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S27 - Take off immediately all contaminated clothing. |
| UN IDs | 1759 |
| WGK Germany | 3 |
| FLUKA BRAND F CODES | 10 |
| HS Code | 29319090 |
| Hazard Class | 8 |
| Packing Group | III |
Formylmethyltriphenylphosphonium chloride - Introduction
(formylmethyl)triphenylphosphonium chloride is an organic compound with the chemical formula [(C6H5)3PCH2]Cl and a molecular weight of 365.43g/mol. The following is a description of the properties, uses, preparation and safety information of the compound:
Nature:
(formylmethyl)triphenylphosphonium chloride is a colorless to yellow crystalline powder. It is stable in air but decomposes when exposed to moisture. This compound is typical of phosphonium chloride salts in which phosphonium (Ph3P) is an organophosphine ligand. It is soluble in a variety of organic solvents, such as ether, methylene chloride and methanol.
Use:
(formylmethyl)triphenylphosphonium chloride are often used as formyl carrier in organic synthesis. It can be used to transfer formyl groups to other molecules, and is commonly used in the reduction of ketones, the carbonylation of thiols, and the deetherification of intramolecular phenols. In addition, it can also be used for the synthesis of organophosphorus compounds and organometallic complexes.
Preparation Method:
(formylmethyl)triphenylphosphonium chloride are generally prepared by reacting the phosphonium salt triphenylphosphonium chloride (Ph3PCl) with formaldehyde or acetaldehyde. The reaction conditions are generally carried out under an inert atmosphere.
Safety Information:
The use or operation (formylmethyl) of triphenylphosphonium chloride requires compliance with appropriate laboratory safety regulations. Since the compound is easily decomposed in a humid environment, it is preferably stored in a dry environment. It is irritating to the skin, eyes and mucous membranes and may be harmful to the respiratory tract. Appropriate personal protective measures should be taken during operation, such as wearing gloves, goggles and protective masks. When handling the compound, avoid inhaling its dust, avoid skin contact and eye contact. In the event of inadvertent exposure to the compound, immediately flush the affected area with plenty of water and seek medical help.
Nature:
(formylmethyl)triphenylphosphonium chloride is a colorless to yellow crystalline powder. It is stable in air but decomposes when exposed to moisture. This compound is typical of phosphonium chloride salts in which phosphonium (Ph3P) is an organophosphine ligand. It is soluble in a variety of organic solvents, such as ether, methylene chloride and methanol.
Use:
(formylmethyl)triphenylphosphonium chloride are often used as formyl carrier in organic synthesis. It can be used to transfer formyl groups to other molecules, and is commonly used in the reduction of ketones, the carbonylation of thiols, and the deetherification of intramolecular phenols. In addition, it can also be used for the synthesis of organophosphorus compounds and organometallic complexes.
Preparation Method:
(formylmethyl)triphenylphosphonium chloride are generally prepared by reacting the phosphonium salt triphenylphosphonium chloride (Ph3PCl) with formaldehyde or acetaldehyde. The reaction conditions are generally carried out under an inert atmosphere.
Safety Information:
The use or operation (formylmethyl) of triphenylphosphonium chloride requires compliance with appropriate laboratory safety regulations. Since the compound is easily decomposed in a humid environment, it is preferably stored in a dry environment. It is irritating to the skin, eyes and mucous membranes and may be harmful to the respiratory tract. Appropriate personal protective measures should be taken during operation, such as wearing gloves, goggles and protective masks. When handling the compound, avoid inhaling its dust, avoid skin contact and eye contact. In the event of inadvertent exposure to the compound, immediately flush the affected area with plenty of water and seek medical help.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: (Formylmethyl)triphenylphosphonium Chloride Visit Supplier Webpage Request for quotationCAS: 62942-43-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (triphenylphosphoranylidene)acetaldehyde Request for quotation
CAS: 62942-43-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 62942-43-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: (Formylmethyl)triphenylphosphonium chloride Request for quotation
CAS: 62942-43-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 62942-43-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: (Formylmethyl)triphenylphosphonium Chloride Visit Supplier Webpage Request for quotationCAS: 62942-43-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (triphenylphosphoranylidene)acetaldehyde Request for quotation
CAS: 62942-43-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 62942-43-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: (Formylmethyl)triphenylphosphonium chloride Request for quotation
CAS: 62942-43-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 62942-43-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
View History


